

Diopside, Inosilicate Structures, Glaucophane in Schist Image Credits – Robert M. Lavinsky CC-BY-SA-3.0, The Assay House, James St. John CC BY-SA 2.0
Inosilicates, commonly known as chain silicates, form one of the most widespread and geologically important silicate groups. Their defining characteristic is the presence of polymerised SiO₄ tetrahedra arranged in chains. These chains can occur in two major forms: single chains, producing the SiO₃²⁻ repeating unit, and double chains, which generate the more complex Si₄O₁₁⁶⁻ structure. The geometry and bonding of these silicate chains create minerals with distinctive cleavage, crystal habit, and physical properties – most notably, the strong prismatic forms and characteristic two-direction cleavage of pyroxenes and amphiboles.
Atomic Structure
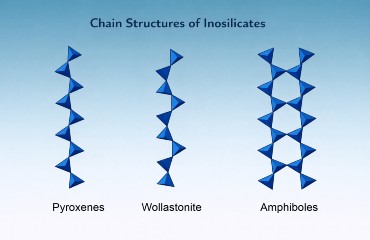
The fundamental building block of inosilicates is the SiO₄ tetrahedron, but unlike isolated or paired structures, these tetrahedra share two oxygen atoms each, linking repeatedly to form extended chains.
Single Chains (Pyroxenes)
Each tetrahedron shares two oxygens with adjacent tetrahedra in a zig-zag pattern, producing an infinite linear chain with the formula (SiO₃)ₙ. These chains bond with metal cations such as Ca²⁺, Fe²⁺, Mg²⁺, and Al³⁺, forming the diverse pyroxene group.
Double Chains (Amphiboles)
In double-chain inosilicates, two parallel chains link together periodically by sharing additional oxygen atoms, forming bands of tetrahedra with the formula (Si₄O₁₁)ₙ. The added polymerization increases structural complexity and creates the basis of the amphibole group, which also incorporates hydroxyl (OH) groups.
These structural differences strongly influence cleavage: pyroxenes cleave at about 90°, while amphiboles cleave at about 56° and about 124°.
Formation Environments
Inosilicates form under a wide variety of geological conditions and are among the most abundant minerals in the Earth’s crust and mantle.
1. Igneous Environments
Pyroxenes are primary minerals in mafic and ultramafic igneous rocks such as basalt, gabbro, and peridotite. High-temperature crystallisation and magnesium-rich melts favour the formation of enstatite, diopside, and augite. Amphiboles may also occur in igneous rocks, particularly in more hydrous magmas such as diorites and andesites.
2. Metamorphic Rocks
Amphiboles, especially hornblende and tremolite–actinolite, commonly form during regional metamorphism. They are key constituents of amphibolite, blueschist, and greenschist facies rocks. Pyroxenes such as omphacite appear in high-pressure metamorphic terranes, including eclogites.
3. Hydrothermal Systems
Hydrothermal alteration can transform pyroxenes into amphiboles or chlorite-bearing assemblages. In metasomatic zones, calcic amphiboles and pyroxenoids may develop in response to fluid–rock interaction.
Examples of Inosilicates
Pyroxenes (single-chain)
Enstatite MgSiO₃
Diopside CaMgSi₂O₆
Augite (Ca,Na)(Mg,Fe,Al)(Si,Al)₂O₆
Amphiboles (double-chain)
Hornblende – a complex, widely distributed amphibole.
Tremolite – Actinolite series Ca₂(Mg,Fe)₅Si₈O₂₂(OH)₂
Glaucophane Na₂Mg₃Al₂Si₈O₂₂(OH)₂ – a key mineral in blueschist.
Uses of Inosilicates
Inosilicates serve crucial roles in geology, industry, and gemology:
Petrological indicators: Pyroxenes and amphiboles are essential for interpreting igneous and metamorphic histories, especially temperature, pressure, and fluid conditions.
Gemstones: Jadeite (a pyroxene) forms one half of the prized gem jade; nephrite (an amphibole) forms the other.
Industrial materials: Pyroxenes contribute to ceramics and refractories due to their stability at high temperatures.
Geodynamic significance: Their presence in mantle rocks helps constrain models of mantle composition and tectonic processes.
Conclusion
Inosilicates are among the most important silicates in geology because their single- and double-chain structures underpin two major rock-forming families – pyroxenes and amphiboles – that record how temperature, pressure, and water content shape igneous and metamorphic rocks. Their chain architecture is also directly reflected in their physical behaviour, especially their characteristic cleavages, making inosilicates a clear example of how atomic arrangement controls mineral properties.
For collectors, inosilicates are rewarding for both variety and locality interest. Pyroxenes can form crisp, dark prismatic crystals in basalts and skarns, while amphiboles range from lustrous hornblende to delicate sprays and fibrous habits in Alpine fissures and metamorphic terrains. The group also includes highly desirable collector and gem materials – most famously jadeite and nephrite (jade) – alongside rarer showpieces such as glaucophane from blueschists or diopside and hedenbergite from skarns. With their strong links to key rock types, wide range of crystal forms, and the chance to build a collection that tells a coherent geological story, inosilicates make an excellent “core” section in any mineral cabinet.
If you are interested in adding specimens of inosilicate minerals to your collection, click HERE.



